|
1/5/2023 0 Comments Period 4 elements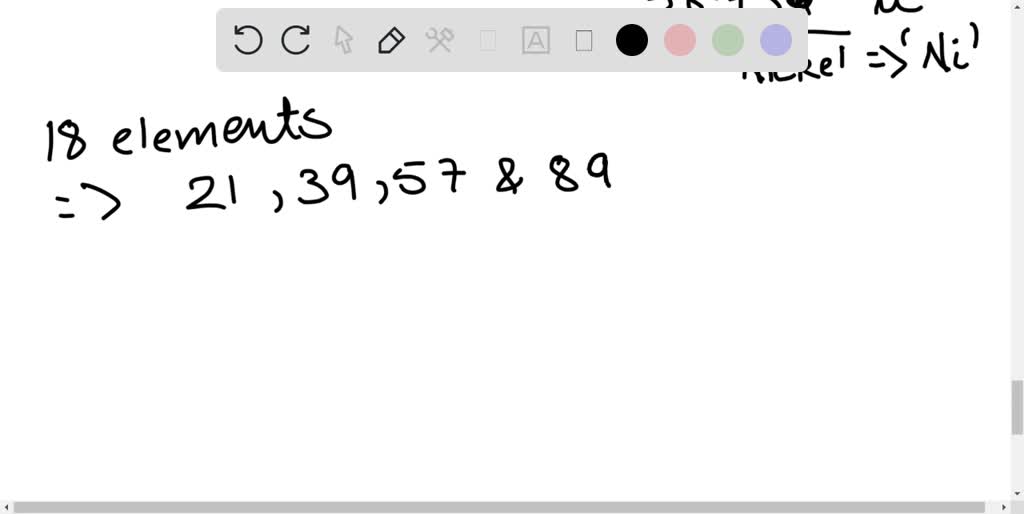
Group 17 has a charge of -1, Group 16 has a charge of -2, Group 15 has a charge of -3. Group 1 elements generally have a charge of +1 when they form ions, Group 2 has a charge of +2, Group 13 has a charge of +3. Group 4A (or IVA) of the periodic table includes the nonmetal carbon (C), the metalloids silicon (Si) and germanium (Ge), the metals tin (Sn) and lead (Pb), and the yet-unnamed artificially-produced element ununquadium (Uuq). And so for an electron configuration for the elements in the third period, so this would be the first period, second period, the third period. 
So 2p6 brings you all the way over to neon. Simply so, what is the name of the element that is in Group 4a and Period 4? For example, this would be 1s1, 1s2 and then 2s2 would be here and then we had six. The period 4 transition metals are scandium (Sc), titanium (Ti), vanadium (V), chromium (Cr), manganese (Mn), iron (Fe), cobalt (Co), nickel (Ni), copper (Cu), and zinc (Zn).īeside above, what element is in group 18 Period 4? Chemically, helium behaves like a noble gas, and thus is taken to be part of the group 18 elements. Similarly, you may ask, what is period 4 on the periodic table? The group consists of nitrogen (N), phosphorus ( P), arsenic (As), antimony ( Sb), bismuth (Bi), and moscovium (Mc). Nitrogen group element, any of the chemical elements that constitute Group 15 (Va) of the periodic table. 
What element is in period 4 Group 15? What element is in period 4 Group 15?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
